>
>
Mornings Matter: Why the Time of Day Could Make or Break Participant Feedback in Clinical Trials
Mornings Matter: Why the Time of Day Could Make or Break Participant Feedback in Clinical Trials
I’ve noticed something about myself: mornings are my sharpest hours. By lunchtime I’m still productive, but after 3pm, focus starts slipping. Evenings? Let’s just say my brain would rather be anywhere else than answering detailed questions.

I’ve noticed something about myself: mornings are my sharpest hours. By lunchtime I’m still productive, but after 3pm, focus starts slipping. Evenings? Let’s just say my brain would rather be anywhere else than answering detailed questions.
That got me thinking: if I know this about myself, why aren’t clinical trials designed around these natural rhythms?
We invest heavily in recruitment, endpoints, and technology, but rarely ask: when are participants most likely to give high-quality feedback?
1. The Science of Human Rhythms
Decades of chronobiology research show that cognitive performance follows circadian rhythms. For most adults:
Morning → peak focus, attention, and memory.
Afternoon → a dip, with slower reaction times and more errors.
Evening → lowest productivity, especially for tasks requiring concentration.
As Valdez (2012) notes, neuropsychological performance is strongly time-of-day dependent, with clear implications for any setting that relies on accurate recall and attention【https://www.dovepress.com/circadian-rhythms-in-cognitive-performance-implications-for-neuropsych-peer-reviewed-fulltext-article-CPT】.
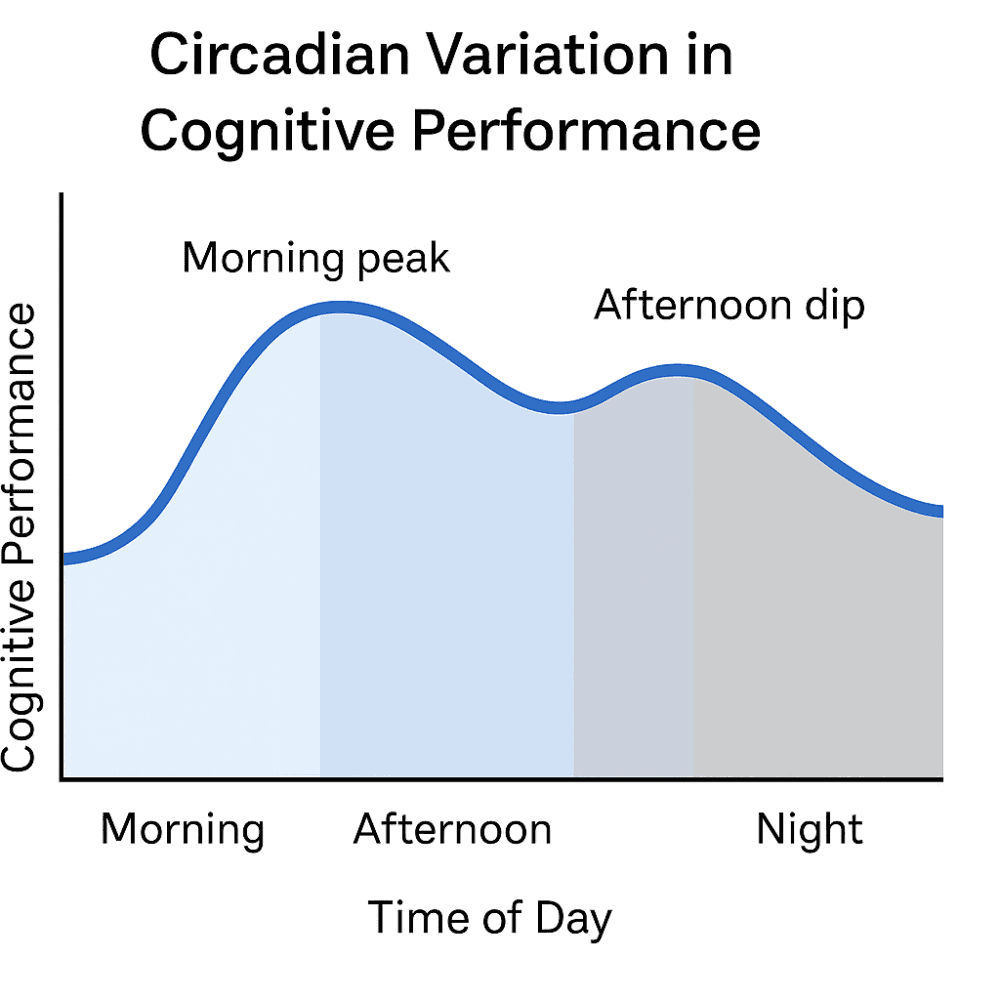
Figure 1: Circadian variation in cognitive performance: attention and memory peak in the morning, dip after lunch, and decline further in the evening (Valdez, 2012).
2. Trials Risk Low-Quality Data
Patient-reported outcomes (PROs) and electronic diaries rely heavily on participant discipline. Studies of ePRO use in rheumatology trials show that while electronic diaries improve timeliness compared with paper, reporting windows still matter — with evening entries more prone to delays, backfilling, and lower compliance (Bingham et al., 2019)【https://pmc.ncbi.nlm.nih.gov/articles/PMC6431038】.
If we nudge participants at the wrong times, we risk:
Higher dropout from “prompt fatigue.”
Lower accuracy in recall or ratings.
Biased data that doesn’t reflect real experiences.
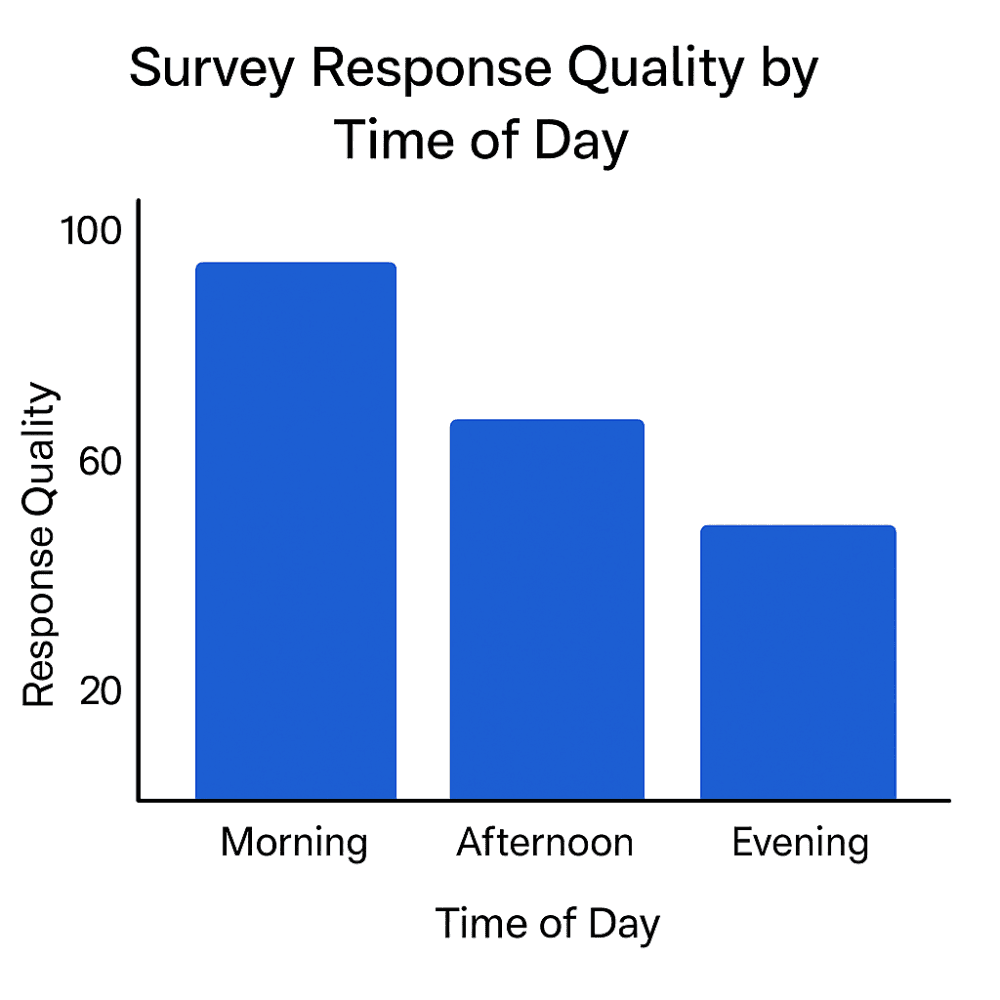
Figure 2: Survey response quality differs by time of day: morning entries are most timely and accurate, while evening reports show higher delays and inconsistencies (Bingham et al., 2019).
3. The Opportunity for Smarter Timing
Instead of fixed daily prompts, why not optimise around when participants are naturally sharper?
Morning-first prompts for general surveys and symptom diaries.
Flexible scheduling based on chronotype (morning vs evening people).
eCOA platforms that adapt, so participants feel supported, not hassled.
Recent work on chronotype and cognitive rhythms shows clear differences in speed and accuracy depending on whether someone is an early or late type (Marchesano et al., 2025)【https://www.mdpi.com/2624-5175/7/1/7】. Designing trials that take these individual patterns into account could reduce burden and improve data quality.

Figure 3: Personalised survey reminders aligned to chronotype: morning types engage better with early prompts, evening types with later ones (Marchesano et al., 2025).
The Takeaway
We’ve spent years talking about “patient centricity.” But are we overlooking one of the simplest ways to respect participants? By asking the right question at the right time.
Maybe the next frontier in clinical trial design isn’t another new device—but simply listening to our body clocks.
References
Valdez, P. (2012). Circadian rhythms in cognitive performance: Implications for neuropsychological assessment. ChronoPhysiology and Therapy, 2, 81–92. Available at: https://www.dovepress.com/circadian-rhythms-in-cognitive-performance-implications-for-neuropsych-peer-reviewed-fulltext-article-CPT
Bingham III, C. O., et al. (2019). Use of daily electronic patient-reported outcome (PRO) diaries in clinical trials: Lessons from rheumatology studies. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC6431038
Marchesano, M., et al. (2025). Circadian rhythms, regular exercise, and cognitive performance. International Journal of Environmental Research and Public Health, 7(1), e7. Available at: https://www.mdpi.com/2624-5175/7/1/7









