>
>
The Protocol Problem: How AI and Automation Can Make Us Better “Conductors” of Our Trials
The Protocol Problem: How AI and Automation Can Make Us Better “Conductors” of Our Trials
If clinical research were an orchestra, the protocol would be its sheet music. Every visit, procedure, and data point flows from its score. Yet, ask any operations lead where delays begin, and you’ll hear the same refrain: the protocol.

When the Blueprint Becomes the Bottleneck
If clinical research were an orchestra, the protocol would be its sheet music. Every visit, procedure, and data point flows from its score. Yet, ask any operations lead where delays begin, and you’ll hear the same refrain: the protocol.
Once a lean guide to a study, it has become a 100-page behemoth: copied, redlined, and reformatted until its logic barely fits together. Every change to an endpoint or schedule echoes through dozens and often hundreds of dependent documents, from consents to CRFs. The result? A process that’s reactive rather than strategic, mechanical rather than musical.
So, what happens when you teach that orchestra to play in sync again?
That’s the question for this edition of The eClinical Edge. And to answer it, I invited Angie Schwab, Founder and CEO of Trialynx™, who recently took the stage at the Whale Tank Showcase to discuss AI’s role in fixing one of clinical research’s most persistent inefficiencies: protocol development.
The Challenge: Complexity Without Harmony
For decades, protocol authoring has been treated as a writing exercise rather than a design discipline. A medical writer drafts; 30 reviewers edit; redlines multiply. By the time version 10 rolls around, no one is entirely sure which version is “final.” Compounding these iterations is the fact that much of this occurs by a “team” of managers who are remotely located and rarely, if ever, come together simultaneously in review.
“We’re at a point where protocols are sophisticated in science but fragmented in structure,” Angie says. “We spend more time formatting decisions than making them.”
This fragmentation has consequences. A 2025 BMC Medical Research Methodology study found that trial protocols have grown more complex each year, yet efficiency and data quality have not kept pace. Without a shared, digital foundation, operational alignment is nearly impossible.
And while every team wants optimisation, the process itself works against it. As Angie puts it:
“We’ve become masters at fixing problems downstream instead of designing them out upstream.”
The Opportunity: Bringing Intelligence to Design
AI and automation are now stepping in where manual coordination has failed. The concept is simple but powerful: capture the logic of a study once: its objectives, populations, endpoints, and visits; then let automation carry those decisions through every downstream document.
Imagine defining a dosing schedule once and watching it populate, in perfect harmony, across the protocol, consent, CRF, and monitoring plan. No duplication, no inconsistency, no rework.
Recent studies suggest this isn’t hypothetical. Researchers Shin et al. (2024) demonstrated that automated generation of schedules of activities reduced manual input and error rates significantly. Olawade et al. (2025) went further, concluding that AI-based trial design tools consistently shorten authoring cycles and improve quality.
Angie describes it as the difference between typing and composing:
“The goal isn’t to replace people; it’s to free them. AI can draft, but humans design. When automation propagates those design choices consistently, teams can focus on strategy rather than syntax.”
Where Regulation Meets Reinvention
This movement isn’t happening in a vacuum. Regulators are quietly re-engineering how study designs are structured and shared.
The ICH M11 guideline introduces a harmonised, machine-readable protocol format that enables consistent data exchange across systems. Similarly, CDISC’s C-SHARE and the Unified Study Definitions Model (USDM) are building the metadata backbone for interoperable study design.
Platforms like Trialynx™ are aligning their frameworks to these standards, turning abstract policy into practical application. In other words, M11 defines the notes; automation ensures the orchestra plays them in tune.
“Digitising the protocol is no longer optional,” Angie notes. “It’s becoming the baseline expectation for transparency and interoperability.”
The Transition: From Documents to Decisions
The biggest challenge isn’t technology, it’s transformation. As organisations modernise, Angie outlines four key areas that help them navigate the change successfully:
Mindset: Shifting from “document-first” to “decision-first” thinking. Protocols are no longer static files; they are dynamic, collaborative models that drive every downstream activity.
Legacy systems: Letting go of patchwork processes built on MS Word® templates and endless email loops, and embracing structured, digital collaboration that connects everyone in real time.
Compliance and governance: Building trust through transparency: ensuring audit trails, role-based access, and traceability are seamlessly embedded within the workflow, not added as an afterthought.
Change management: Empowering medical writers, clinicians, and scientists to focus on their expertise: guiding study design, interpreting data, and shaping strategies, rather than formatting, reconciling, and version tracking.
These shifts signal more than operational maturity they represent a philosophical change. Protocol development moves from a static documentation process to a dynamic system of decision-making.
Teams that have embraced this shift report fewer amendments, shorter cycle times, and clearer communication across functions. Not because AI thinks for them, but because it clears the noise so humans can focus on insight.
“People often think automation will depersonalise the process,” Angie says. “It does the opposite. It gives us time back to think, to connect, to design.”
The Future: From Paper to Living System
The protocol of tomorrow won’t be a static document. It will be a living, learning framework, a digital model that evolves with each decision, integrates regulatory standards by default, and feeds directly into downstream systems.
As Angie puts it, “We’re moving from protocol as paperwork to protocol as a data model.”
In that world, AI won’t replace clinical experts; it will amplify them. Automation will handle alignment; scientists will handle insight. The result is a trial design process that’s faster, cleaner, and more patient-focused than ever before.
Conclusion: Time to Play in Sync
For too long, the industry has accepted inefficiency as inevitable. But with AI and automation now maturing and with frameworks like ICH M11 and CDISC C-SHARE setting the standard the opportunity to redesign trial development is here.
As Schwab reminds us, innovation doesn’t start in the lab; it starts in the plan. When we align science, systems, and standards from the very first draft, we don’t just build better trials, we build them in harmony.
About Angie Schwab MS, PMP: a clinical research futurist with over 20 years of experience in clinical trial design, healthcare automation, and AI-driven innovation.

Angie Schwab - on stage at the recent Whale Tank Showcase.
For more on Trialynx™: https://www.trialynx.io/about
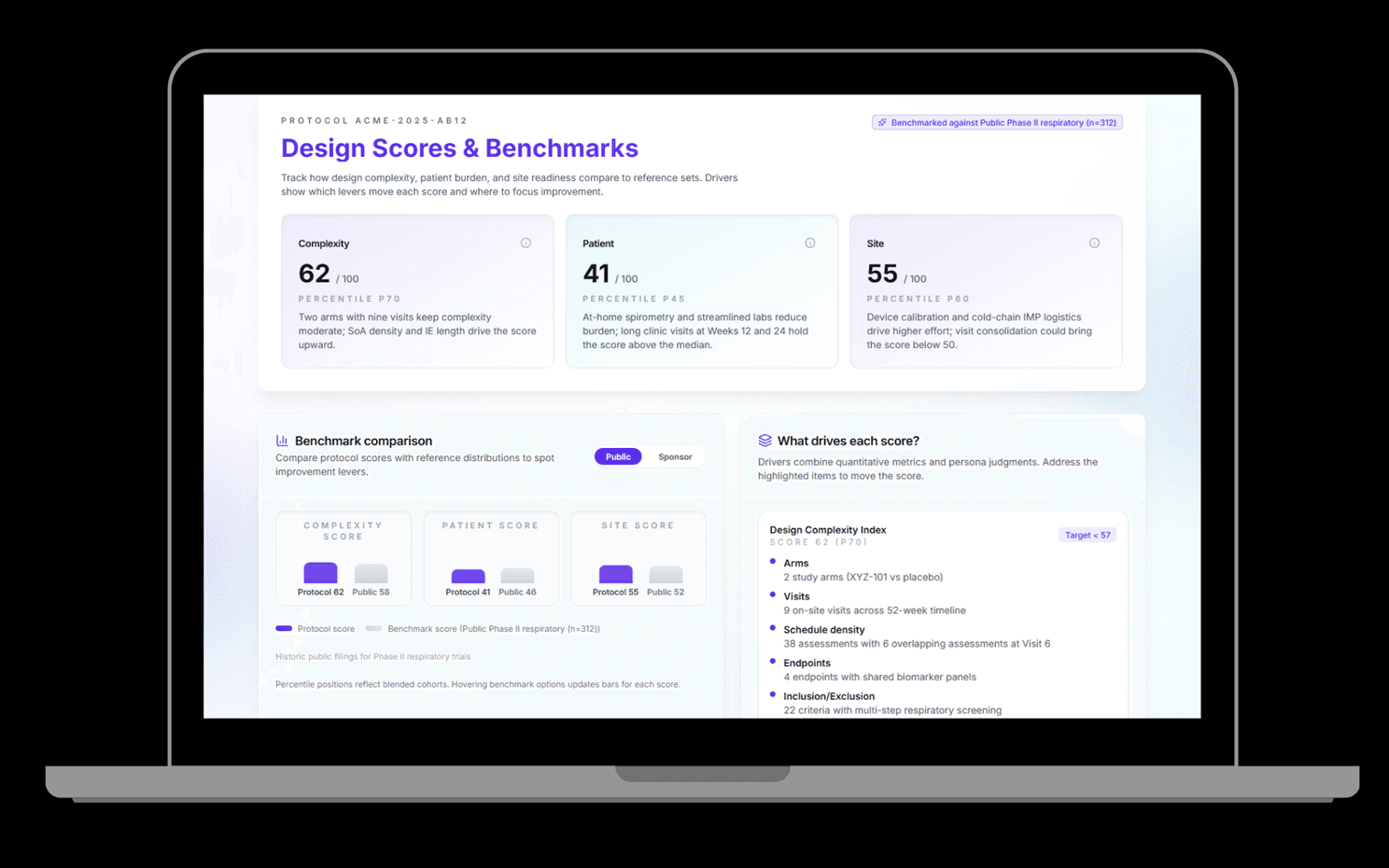
Trialynx software
References
Willigers BJA et al. (2025). Development of a Protocol Complexity Tool (PCT): Clinical trials are becoming more complex. BMC Medical Research Methodology. https://bmcmedresmethodol.biomedcentral.com/articles/10.1186/s12874-025-02652-9
Shin K et al. (2024). Automated Protocol Templates with Efficient Schedule of Activities Table Generation in Healthy Volunteer Trials. SSRN. https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5033742
Olawade DB et al. (2025). Artificial Intelligence in Clinical Trials: A Comprehensive Review. Elsevier. https://www.sciencedirect.com/science/article/pii/S1386505625003582
Teodoro D et al. (2025). A Scoping Review of Artificial Intelligence Applications in Clinical Trials. npj Digital Medicine. https://www.nature.com/articles/s41746-025-01886-7
ICH M11 Guideline. International Council for Harmonisation. https://www.ich.org/page/m11-protocol
CDISC C-SHARE and USDM Overview. Clinical Data Interchange Standards Consortium. https://www.cdisc.org









