The Silent Shift: Why CROs Are Quietly Rebuilding Their Tech Stacks
Something curious is happening across the CRO landscape. Not loud. Not headline-grabbing. No “revolutionary platform announcements” or glossy product splash pages. Instead: a quiet, deliberate dismantling of internal technology ecosystems followed by a rebuild.

Something curious is happening across the CRO landscape. Not loud. Not headline-grabbing. No “revolutionary platform announcements” or glossy product splash pages.
Instead: a quiet, deliberate dismantling of internal technology ecosystems followed by a rebuild.
CROs are restructuring their tech stacks for 2026. Not to look innovative. But to stay operational.
This is not a platform war. This is an architectural correction.
Innovation Without Integration Is Just Decor
For years CROs collected technology like people collect apps: “Try this one,” then “just one more,” until the ecosystem resembles a digital junk drawer.
By 2024, many CROs were simultaneously running:
more than one EDC,
multiple IRTs chosen by individual study teams,
eCOA solutions bought for speed rather than fit,
bespoke internal tools patched together with goodwill, macros, and prayer.
It functioned. Technically. But like a 20-year-old toaster it worked right until the day it didn’t.
Research supports this tension. npj Digital Medicine notes that digital tools absolutely improve trial efficiency but only when the underlying data models and systems play well together (Mittermaier et al., 2023). In other words: digital innovation doesn’t matter if your pipes can’t carry the water.
Hence the rebuild.
What’s Driving This Silent Renovation?
1. The CRO model itself is changing
Boston Consulting Group’s recent analysis of CROs (2024) paints a clear picture: the traditional model — “throw people at the problem and hope the systems keep up” — is dying.
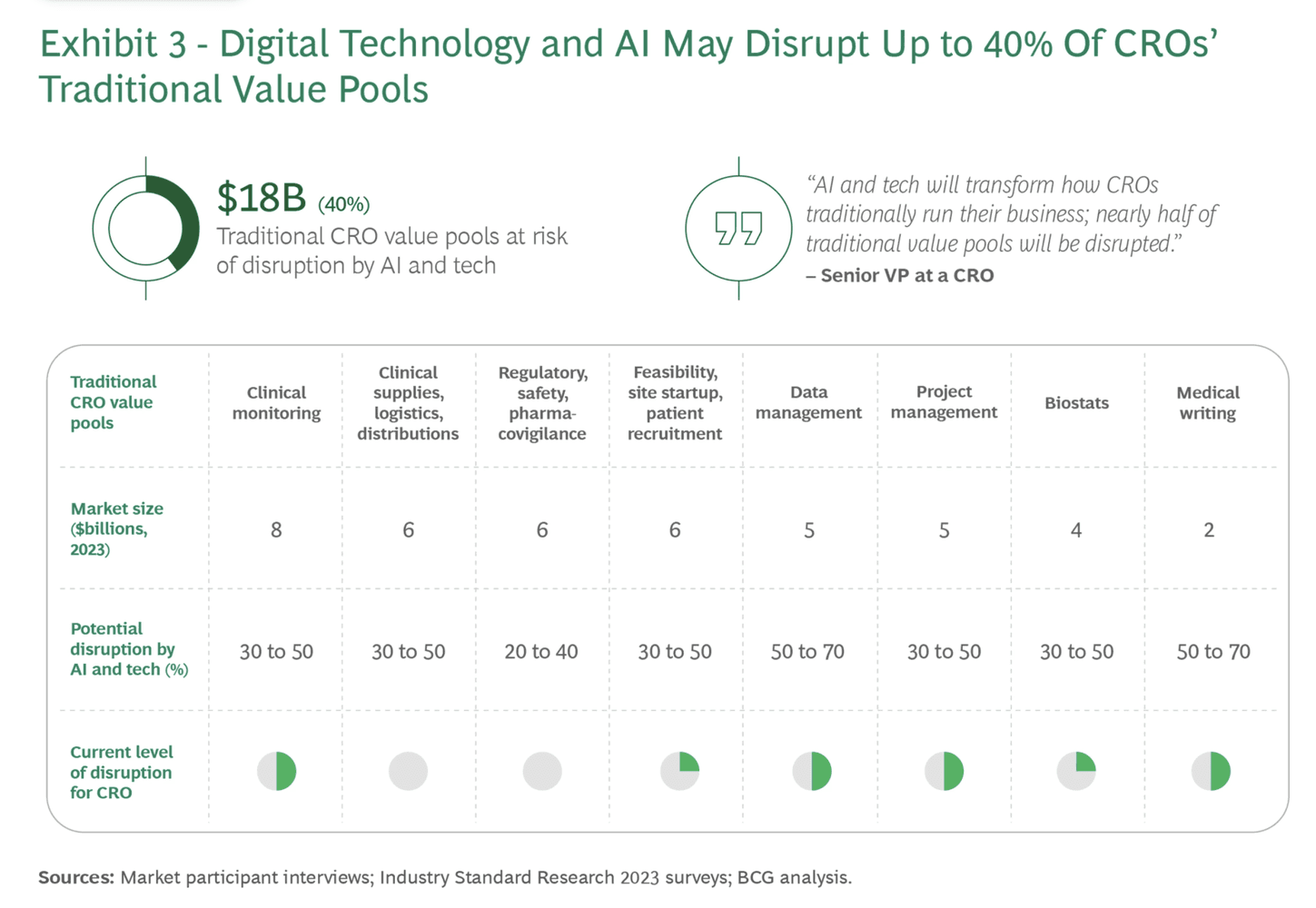
Exhibit 3: The shift from transactional CRO delivery to integrated, tech-enabled operating models. Source: BCG
Sponsors increasingly choose partners not on the depth of their slide decks but the quality of their tech operating model: integration, data lineage, configurability, and speed of change.
2. Sponsors want consistency, not a digital buffet
RFPs are no longer asking “What system do you use?” They’re now asking:
“Show us your architecture.”
“Show us your integrations.”
“Show us how protocol → database mapping is automated.”
“Show us your AI governance.”
That’s not vendor hype. That’s a structural expectation.
And CROs with sprawling, inconsistent tech stacks are losing ground before they’ve even written the first line of their proposal narrative.
3. Frankenstacks have reached their limits
If you’ve ever seen a CRO tech diagram that looks like a cross between a subway map and a bowl of spaghetti, you know what I mean.
The feasibility study published in the Journal of Medical Internet Research (Harrison et al., 2024) makes this painfully clear: digital solutions improve trial speed only when they’re deployed in a consistent, integrated way. Fragmentation isn’t an annoyance it’s an operational hazard.
The End of the Bolt-On Era
The industry has hit a turning point: You cannot keep bolting “shiny new digital tools” on top of architecture from 2010 and expect them to behave.
2025 is the year many CROs quietly admit this.
Enter the era of:
simplified stacks,
cleaner data models,
well-defined ownership,
standardised workflows,
fewer vendors but deeper capability.
This is not flashy innovation. It’s groundwork, the kind that accelerates everything else.
What Sponsors Will Feel in 2026
The CRO proposals of 2026 will look different. Expect to see:
fewer vendor logos,
more architectural maps,
clearer data lineage,
stronger integration stories,
protocol-to-database workflows,
AI explainability frameworks (because regulators are watching closely).
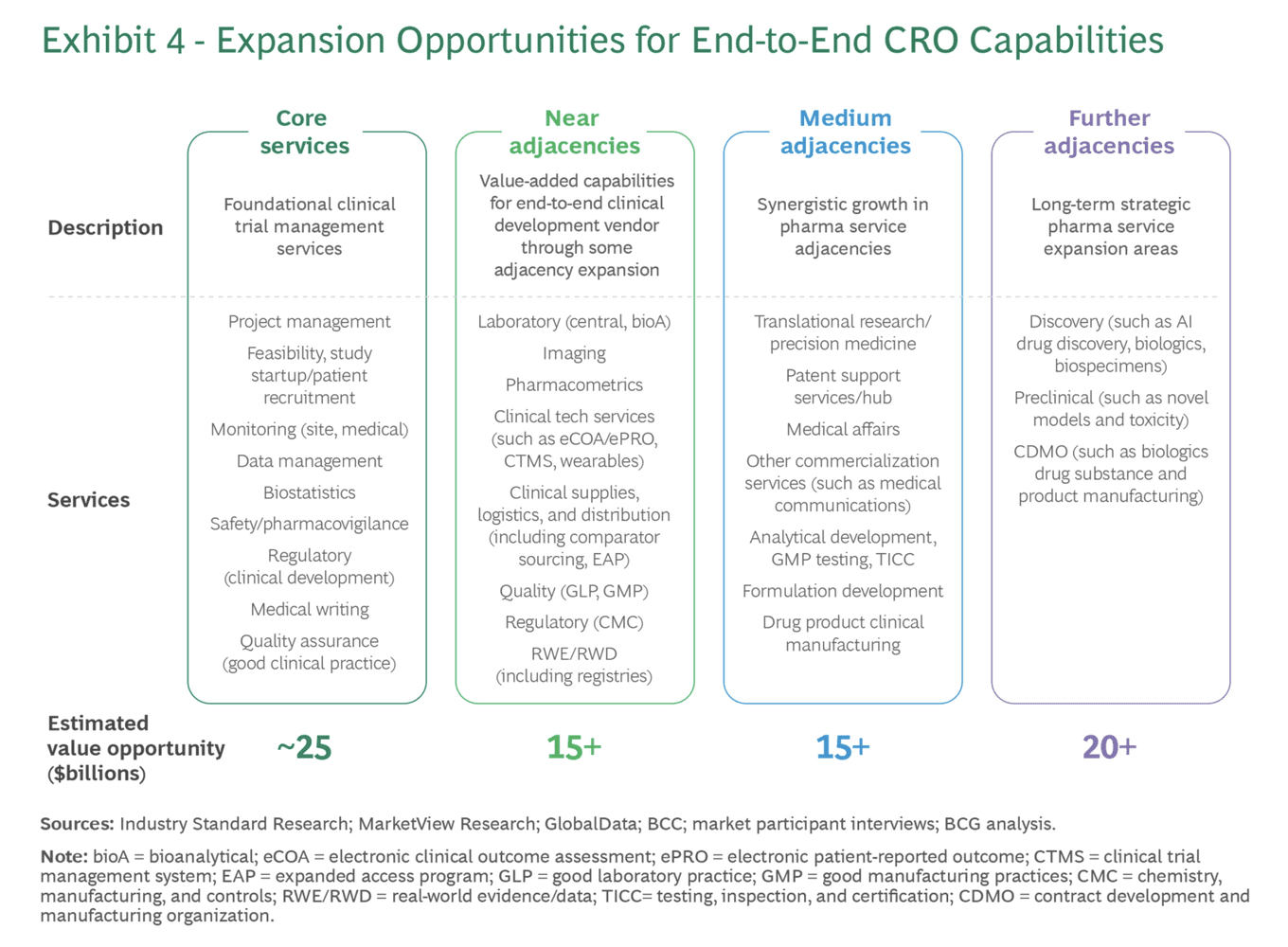
Exhibit 4 shows the capabilities CROs must build. Source: BCG
The message CROs will push: “We can run your trial faster because our stack is coherent.” Not because it’s revolutionary. Because it’s finally functional.
This Isn’t About Tech. It’s About Risk.
CROs aren’t transforming because someone fell in love with a new digital acronym.
They’re transforming because:
fragmented systems introduce study-level risk,
risk slows timelines,
slower timelines cost money.
That’s the chain. That’s the real story.
As BCG highlights, CROs that modernise their technology architecture will become strategic partners. Those who don’t will remain service vendors and increasingly replaceable ones.
It’s a Quiet Revolution But a Revolution Nonetheless
No fireworks. No platform hype. Just a necessary restructuring of the digital foundations that shape the modern clinical trial.
The next competitive advantage won’t be who has the most digital tools. It will be:
Who has the fewest unnecessary ones — connected cleanly — and working in unison.
2025 won’t reward the flashiest stack. It will reward the strictest architecture.
And the CROs who are quietly rebuilding today will be the ones sponsors quietly prefer tomorrow.
References
1. Harrison, T. M. et al. (2024). “Digital Solutions Observed in Clinical Trials: A Formative Feasibility Study.” Journal of Medical Internet Research, 26:e47882. https://doi.org/10.1038/s41746-023-00841-8
2. Boston Consulting Group (2024). “Big Changes Are Coming to Pharma CROs.” https://www.bcg.com/publications/2024/big-changes-to-pharma-cros
3. Mittermaier, M., Venkatesh, K.P., & Kvedar, J.C. (2023). “Digital health technology in clinical trials.” npj Digital Medicine, 6:88. https://www.jmir.org/2024/1/e47882









