TikTok Could Be the Saviour of Modern Clinical Trials...
A question keeps pestering me: why aren’t more people proactively engaging in research?

A question keeps pestering me: why aren’t more people proactively engaging in research?
It’s never been easier to reach people online. Sites and recruitment teams are constantly looking for new participants. So what’s the real blocker?
In this piece I unpack three main reasons why people don’t participate in research, then propose three ways modern technology (yes, including TikTok) can help.
I also ask you the question:
Why are we not doing personalised Electronic Health Record Smartphone prompts in 2025?
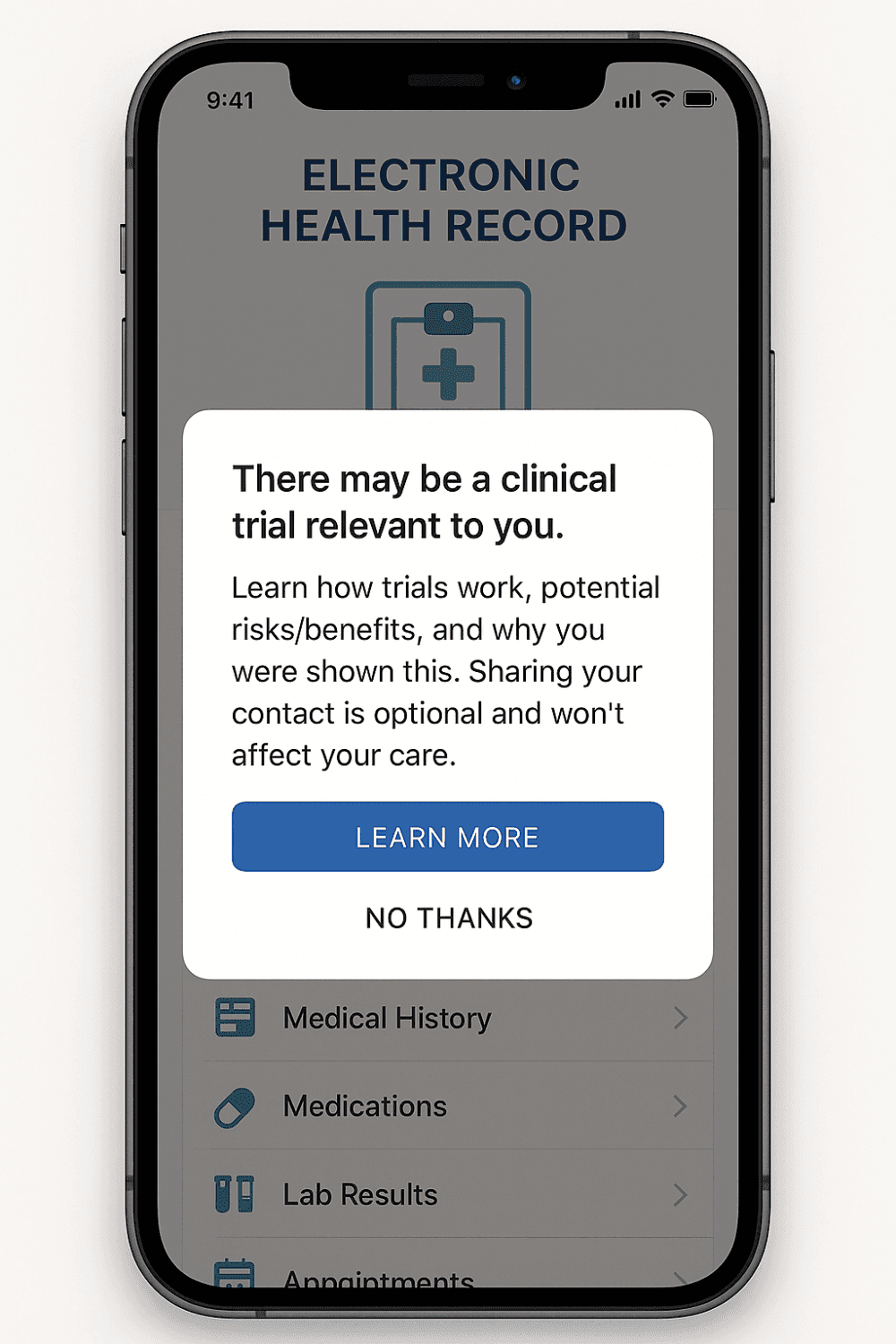
This should be on your phone by now, its not on mine and I don't know why.
Why People Don’t Participate in Clinical Trials
1) Lack of Awareness
Most people simply don’t know trials are even an option. A national survey found that 41% of U.S. adults reported not knowing about clinical trials, with even higher gaps among minority groups [1].
2) Negative Perceptions & Distrust
“Guinea pig” myths, fear of side effects, and mistrust in pharma persist—especially in communities historically under-served by research. Qualitative studies show that distrust, perceived exploitation, and discrimination are key reasons people avoid participation [2].
3) Inconvenience & Burden
Travel, time off work, paperwork, and site visits add up. For many, the perceived hassle outweighs potential benefits. Research on decentralised models shows that reducing burden significantly improves recruitment and retention, particularly for rural and underserved groups [3].
How Modern Technology Can Help (Backed by Data)
A) Smartphones & Social Video: Meet people where they already are
TikTok’s global ad reach is ~1.59 billion people as of January 2025 [4]. In the U.S., one-third of adults use TikTok, rising to 62% of those aged 18–29 [5].
And it’s not just reach; it’s influence. 52% of TikTok users—17% of all U.S. adults—regularly get news on the platform, shaping perceptions and decisions daily [6].
Short, authentic video explainers from clinicians and past participants could demystify trials, bust myths, and funnel people straight into pre-screening.

Between the noise of teen dance crazes and epic cat videos, I'd like to see this.
B) Decentralisation + eCOA: Reduce the hassle, raise retention
If burden is the problem, decentralisation is the solution. Tele-visits, eConsent, and ePRO reduce travel/time costs and expand reach. Regulators now encourage these approaches, and evidence shows they boost retention and diversify participants [3].
C) Smart Targeting for Recruitment
Systematic reviews show that online recruitment often outperforms traditional methods in speed and cost, particularly for hard-to-reach groups [7].
With TikTok and other social platforms, sponsors can use demographic and interest-based targeting to reach potential participants where they already are.
Considerations: “Do IRBs Stifle Social Media?” ** (explained in more detail below)
Regulators don’t prohibit digital or social media for recruitment; they define how to do it responsibly.
FDA: Advertising to research subjects is “not in and of itself objectionable.” Basic trial listings online don’t require IRB pre-approval [8].
ICH GCP: IRBs/IECs should review the description of the media and recruitment ads—meaning social is allowed with oversight [9].
EMA: The ACT-EU paper encourages use of digital tools to “facilitate recruitment” and participant trust [10].
If an IRB demands restrictions beyond what’s required, sponsors can (politely) ask them to link the request to a specific participant risk.
The Silver Bullet: Ethical “Pop-Up” Trial Matching via Public EHR Portals*
Imagine if trial awareness arrived where people already manage their care: their national patient portal app. With explicit opt-in, the app could run a privacy-preserving match against active trials and gently notify both the patient and their treating clinician.
EHR alerts work: Studies show physician-facing alerts significantly increase recruitment [11–13].
Patient portals are viable: Embedding recruitment into portal check-ins has been successfully trialled [14].
FHIR-native matching exists: Prototypes already use FHIR data to compute trial eligibility and explain results [15,16].
The guardrails are clear: strict consent, IRB-approved language, frequency caps to avoid alert fatigue [13], and equity monitoring to ensure all groups benefit.
I've dived deeper on this concept below for those who want to explore it further.*
Conclusion
Recruitment will always be challenging, but maybe it doesn’t need to be quite this hard.
Awareness, perception, and burden are the three biggest barriers. Social media, decentralisation, and smart targeting don’t solve everything, but they could make recruitment less of a constant uphill battle.
And yes, I’m only half-joking when I say: maybe TikTok really could be the saviour of modern clinical trials.
P.s Read on below the reference section if you want to know more about IRB regulations or EHR Smartphone Apps.
References
1. Barriers to Participation
Yadav, S., et al. (2022). Perceptions of and barriers to participation in clinical research: A US national survey. Journal of the Royal College of Physicians of Edinburgh.
Scharff, D.P., et al. (2010). More than Tuskegee: Understanding mistrust about research participation. Journal of Health Care for the Poor and Underserved, 21(3), 879–897.
Goodson, N., et al. (2022). Opportunities and challenges of decentralised clinical trials: a systematic review. BMC Medicine, 20, 124.
2. Social Media & Awareness
DataReportal. (2025). TikTok users, stats and trends.
Pew Research Center. (2024). Americans’ Social Media Use.
Pew Research Center. (2025). A closer look at Americans’ experiences with news on TikTok.
Brøgger-Mikkelsen, M., et al. (2020). Online recruitment of participants for clinical trials: A systematic review. Contemporary Clinical Trials Communications, 19, 100638.
3. EHR, Matching & Regulatory Footing
U.S. FDA. (1998). Recruiting Study Subjects – Information Sheet.
ICH. (2023). E6(R3) Good Clinical Practice – Draft Principles.
EMA & HMA (2022). Recommendation Paper on Decentralised Elements in Clinical Trials.
Embi, P.J., & Leonard, A.C. (2005). Effect of a Clinical Trial Alert System on Physician Participation in Trial Recruitment. JAMA Internal Medicine, 165(19), 2272–2277.
Embi, P.J., et al. (2005). Development of an EHR-based Clinical Trial Alert System. JAMIA, 12(3), 315–321.
Embi, P.J., et al. (2012). Evaluating alert fatigue to EHR-based clinical trial alerts. JAMIA, 19(e1), e145–e148.
Leuchter, R.K., et al. (2023). Embedding research recruitment within the patient portal (preCheck-in). JAMIA.
Alper, B.S., et al. (2023). Striking a Match between FHIR-based patient data and trial eligibility. Learning Health Systems.
SMART Health IT. (2023). Accelerating clinical trial recruitment with Bulk FHIR.
U.S. Department of Health & Human Services. (n.d.). HIPAA: Preparatory to Research.
U.S. Department of Health & Human Services. (n.d.). Research and the HIPAA Privacy Rule.
European Commission. (2019). Q&A: Data Protection and Clinical Trials.
*Ethical “Pop-Up” Trial Matching via Public EHR Portals
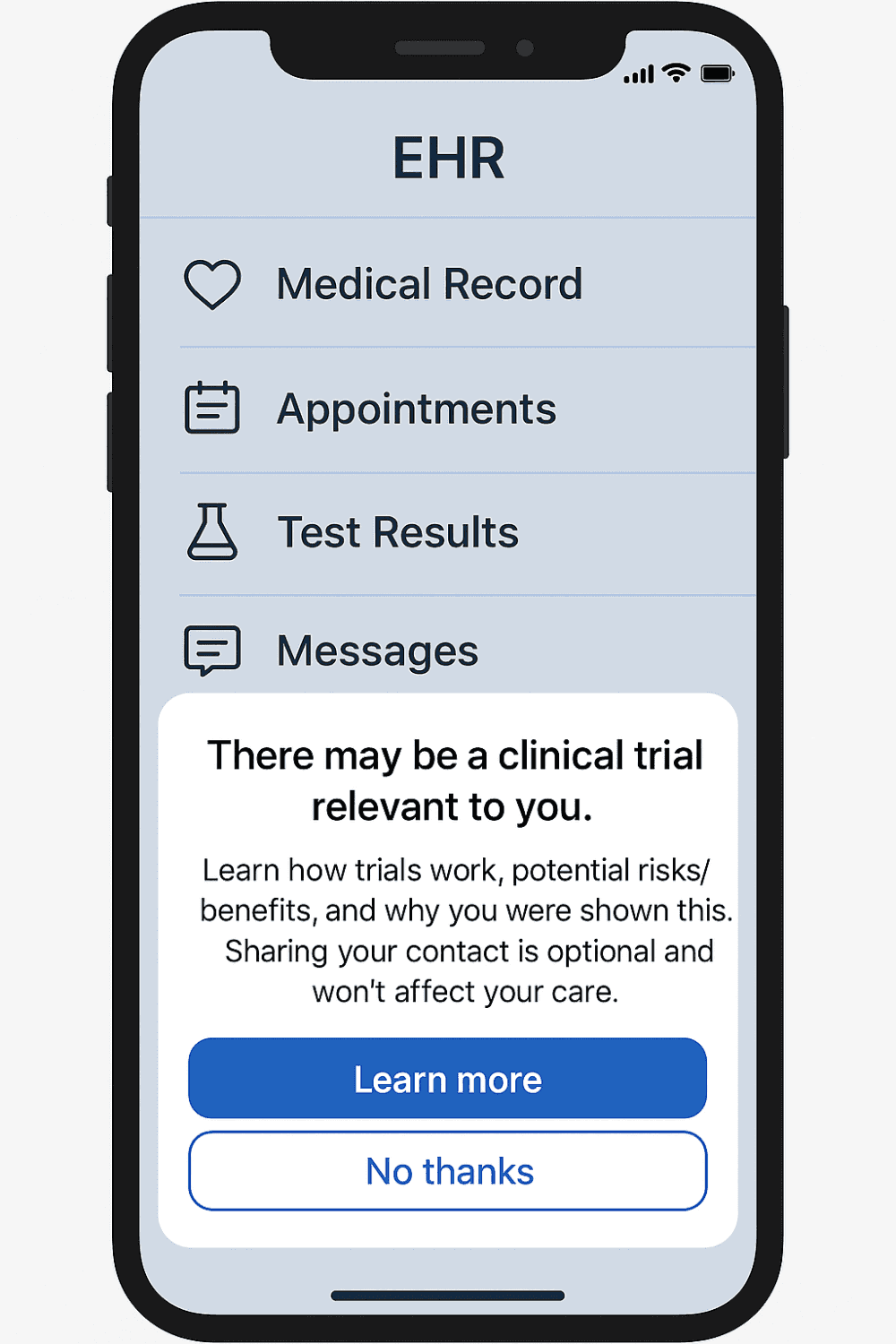
Private or public health service - this technology exists and should be used.
How it would work (architecture in 7 steps)
Government-backed portal, explicit opt-in Public health ministries (or national health services) offer a standard patient portal app. During onboarding, users can opt in to “trial matching.” No opt-in = no matching. Legal backbone (US): HIPAA allows “preparatory to research” activities to identify potential participants without removing PHI; contacting patients still requires proper authorisation or a covered workflow. (HHS.gov, irb.northwestern.edu) EU context: GDPR permits processing for research/public interest under strict safeguards laid out in EC/EMA guidance; transparency and proportionality are key. (Public Health, PMC)
Standards-first data exchange The portal retrieves a minimal, consented data slice (problems/diagnoses, meds, age, labs ranges, location) via HL7 FHIR APIs from the public EHR. Matching engines that already operate fully on FHIR JSON are feasible today. (PMC)
Trial ingestion + computable criteria Active trials (from registries/sponsors) publish machine-readable inclusion/exclusion (e.g., FHIR/CTSA or vendor CDL). A FHIR-native matching service compares criteria to the patient’s profile and returns a simple match score + rationale (“meets age, diagnosis; lab value borderline”). (EHR Good Neighbor)
Dual-channel nudges (patient + clinician) Patient: In-app card: “A clinical trial may be relevant. Learn more / Decline.” Clinician: Non-interruptive EHR inbox message or passive clinical trial alert (CTA) in workflow; proven to increase recruitment when done right. (jamanetwork.com, PMC, academic.oup.com)
Frictionless next steps If the patient taps “Learn more,” they see plain-English details and can share contact details with the site or request their clinician to initiate a referral. No data leaves the covered entity without the right authorisation.
Governance & IRB/Ethics review The ad copy and the channels (portal card language, EHR message) go to IRB/ethics for review; this is squarely within FDA/ICH expectations for recruitment materials and the media used (you already address this in your “Considerations” section).
Audit-ability & safety Full audit logs, frequency caps (e.g., ≤1 suggestion/month), and easy opt-out. Evaluate safety signals (e.g., anxiety from alerts) and equity (matching works across languages and demographics).
Why this is more than theory
Physician-facing EHR alerts work. Randomised/controlled and observational studies show EHR-embedded “clinical trial alerts” increase enrolment and physician participation—the model is proven inside clinical workflow. (jamanetwork.com, PMC, academic.oup.com)
Patient portals can host recruitment. Studies embedding recruitment and authorisation inside portal pre-check-in show operational feasibility and uptake. (PMC)
FHIR-based matching exists. Open, peer-reviewed prototypes match FHIR patient data to computable eligibility and explain results transparently. (PMC)
Guardrails (the hard bits you must get right)
Consent & minimisation: Only use data that are strictly necessary to screen; show what’s used, by whom, and why (HIPAA/GDPR transparency). (HHS.gov, Public Health)
Non-coercive copy: “May be eligible” ≠ “You qualify.” No undue influence; IRB-approved text, consistent with FDA’s view that advertising itself isn’t objectionable but must be factual.
Alert fatigue: Start with passive messages and cap frequency; CTA studies highlight alert fatigue risks if over-used. (academic.oup.com)
Equity & bias: Benchmark match rates by language, region, and demographics; tune criteria and outreach to avoid systematic exclusion.
Metrics that matter
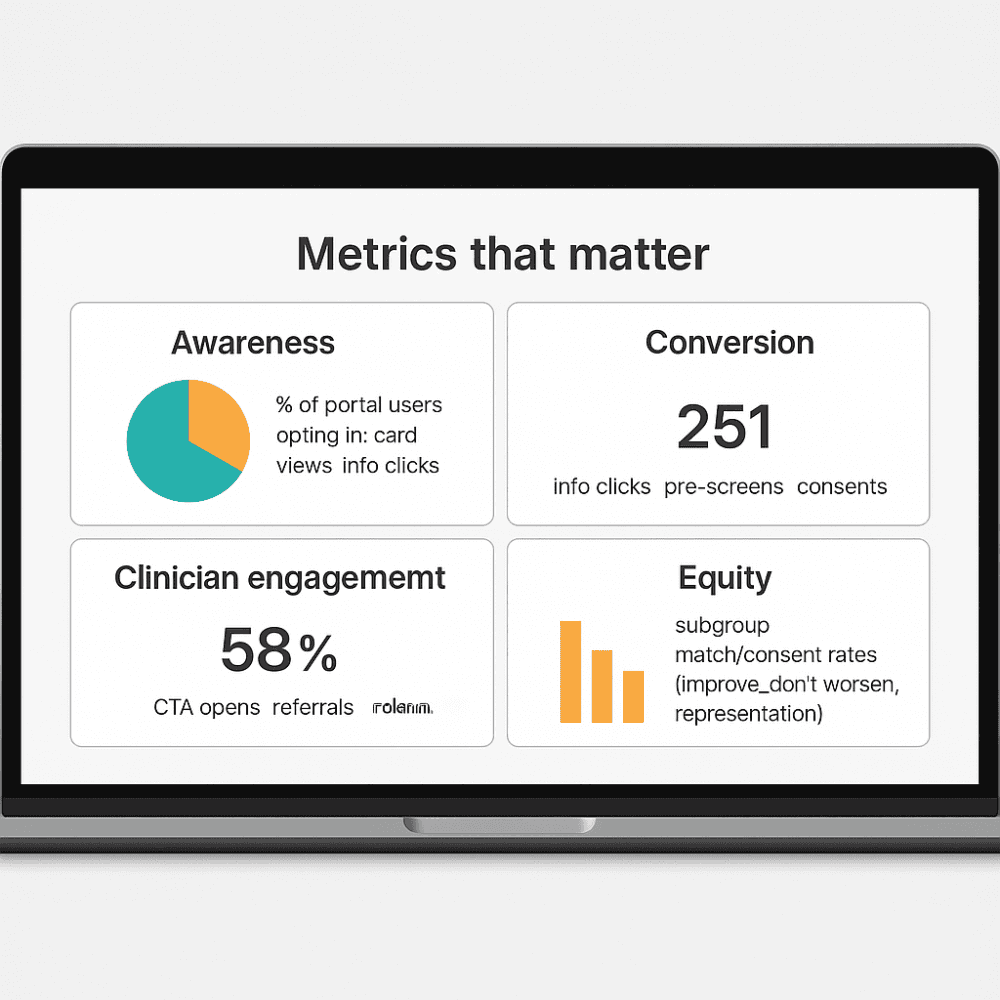
Awareness: % of portal users opting in; card views → info clicks.
Conversion: info clicks → pre-screens → consents.
Clinician engagement: CTA opens → referrals → randomisations.
Equity: subgroup match/consent rates (improve, don’t worsen, representation).
Experience: patient/clinician satisfaction; opt-out reasons.
**Do IRBs Stifle Social Media? Short answer: No: use the rulebook.
Regulators don’t prohibit digital or social media for recruitment; they define how to do it responsibly and what IRBs must review.
FDA (IRB & advertising): FDA states that “direct advertising for research subjects… is not in and of itself, an objectionable practice.” IRBs should review the content and mode of recruiting ads to ensure they’re not coercive or misleading. Importantly, basic clinical-trial listings on the internet (title, purpose, eligibility, sites, contact) do not require prior IRB approval because additional review “would provide no additional safeguard.” (U.S. Food and Drug Administration)
ICH GCP E6(R3): ICH makes this explicit: IRBs/IECs should review “other trial-related information to be provided to trial participants, including the description of the media through which such information will be provided,” plus any “advertisement for participant recruitment (if used).” In other words, digital/social is fine—just ensure IRB review of the materials and channel plan. (European Medicines Agency (EMA))
EMA / EU (DCT recommendation): The EMA/HMA ACT-EU Recommendation Paper on Decentralised Elements recognises “increasing use of digital tools within clinical trials” and explicitly aims to facilitate their use while keeping participant protection paramount. It also encourages early participant involvement to “develop trust… facilitate recruitment, and promote adherence,” and notes that sponsors should transparently summarise decentralised elements for ethics committee assessment—not to ban them, but to assess proportional risk.
How to (politely) push back when an IRB adds extra hurdles
Anchor to ICH: E6(R3) says IRBs/IECs review ads and the media you’ll use; that’s the bar. If an IRB demands restrictions beyond what protects rights, safety, or data integrity, ask them to tie the request to a concrete risk (ICH allows additional info when it “adds meaningfully to the protection” of participants). (European Medicines Agency (EMA))
Cite FDA’s internet-listing carve-out: If you’re posting a basic listing (title, purpose, eligibility, site, contact) on a registry or platform that constrains content, point to FDA’s note that IRB pre-review isn’t required in that case. (U.S. Food and Drug Administration)
Use EMA’s facilitative stance: In the EU, emphasise the ACT-EU goal to enable decentralised/digital approaches with proportional oversight—ethics committees assess suitability and transparency; they’re not instructed to prohibit digital outreach per se.
Bottom line: Social media isn’t the problem; unclear, over-promissory ads are. Keep copy factual and non-coercive, route ad content and channel plan for IRB review (as ICH/FDA expect), and use EMA’s DCT paper to frame digital outreach as good practice with proportional safeguards, not something to be blocked.









