What B2B Sales (Research) Has Taught Me About B2B Sales
Why clinical trial technology buyers and sellers need to step up in 2026 In case you’ve been living under a rock - or buried under a pile of protocols - there’s a meme doing the rounds on LinkedIn and X that goes something like this: “I just had a deeply personal life experience… and here’s what it taught me about B2B sales.”

Why clinical trial technology buyers and sellers need to step up in 2026
In case you’ve been living under a rock - or buried under a pile of protocols - there’s a meme doing the rounds on LinkedIn and X that goes something like this: “I just had a deeply personal life experience… and here’s what it taught me about B2B sales.”

The joke took off after a well-intentioned post (pictured above) likened a marriage proposal to a “Closed Won” opportunity and was promptly roasted for being cringey, self-referential, and meaningless. The meme stuck because it captured something many people in B2B already feel: we’re drowning in performative insight and starving for real ones.
And yet, the joke works because it points at a deeper truth. In B2B tech sales, especially in clinical trial technology, we often default to comforting narratives and tidy analogies instead of confronting how buying and selling actually happens in complex, regulated environments.
So, this isn’t another meme-driven article. It’s an attempt to do the opposite: to use evidence, not anecdotes, to explain why so many clinical trial tech conversations stall and what buyers and sellers need to change in 2026.
The uncomfortable truth: buyers help create the sales behaviour they complain about
Ask sponsors or CROs what frustrates them about technology vendors and you’ll hear some familiar answers:
“They all sound the same.” “Too salesy.” “Too feature-focused.” “They don’t understand our real problem.”
What buyers say — and what the evidence shows
“They all sound the same” / “Too salesy” / “Too feature-focused”
Research from Gartner shows that 73% of B2B buyers actively avoid suppliers who send irrelevant outreach, particularly early in the buying journey, because it feels pushy and low-value. This aligns with broader buyer-behaviour research showing increasing scepticism toward generic, feature-led sales engagement. Buyers prefer to self-serve most of the evaluation process and only engage sellers when they expect insight, not repetition.
“They don’t understand our real problem”
Global B2B buyer experience research shows that over half of buyers believe sellers fail to understand their friction points or business context. This gap is especially visible in clinical trial technology, where vendors often arrive late in the evaluation process - after buyers have already formed preferences - but without sufficient understanding of trial design, operational constraints, or regulatory exposure.
“Too salesy” / “Prefer rep-free experiences”
Gartner also reports that 61% of B2B buyers now prefer a rep-free buying experience, a signal that many find traditional sales engagement intrusive unless it delivers clear, differentiated insight. Buyers frequently delay or avoid engaging sales teams until late in the decision cycle for precisely this reason.
Now here’s the uncomfortable part.
Research into enterprise B2B buying consistently shows that many buying groups engage vendors before they’ve aligned internally on what they actually need. Decisions typically involve 6–10 stakeholders, each with different priorities, incentives, and risk tolerance. In clinical trials, that number is often higher spanning Clinical Operations, Data Management, Biostatistics, IT, QA, Procurement, and sometimes Medical or Digital Innovation.
When buyers invite vendors in before that alignment exists, they create a vacuum.
And vacuums get filled with:
Generic decks
Feature walkthroughs
Safe, non-committal messaging
In other words, the exact sales behaviour buyers say they dislike.
How clinical trial technology buyers really judge vendors (even if they never say it)
Buyers like to say they’re evaluating:
Functionality
Compliance
Integration
Cost
Scalability
They are. But research into complex B2B decision-making shows they’re also asking quieter, more personal questions:
Will this decision increase or reduce my personal risk? (Operational, regulatory, reputational)
Can I defend this choice to QA, IT, and Procurement if something goes wrong?
Will this vendor help me align stakeholders I don’t control?
In clinical trials, technology buying is not just technical - it’s political and professional. Sponsors and CRO leaders are not simply choosing software. They are choosing who absorbs risk when timelines slip, inspections happen, or sites struggle.
That’s why technically strong vendors still lose. Not because they lack features - but because they fail to help buyers manage uncertainty.
What the research actually says about effective B2B selling in regulated industries
Strip away the LinkedIn noise and a few evidence-based patterns appear consistently.
1. Buyers are already deep into the decision journey before sales shows up
Multiple studies show B2B buyers complete a large portion of their evaluation independently before engaging vendors. They’ve read content, spoken to peers, and formed early preferences.
In clinical trial technology, this means:
First meetings are rarely exploratory
They are validation or elimination moments
If early conversations don’t add insight beyond what buyers already know, credibility evaporates quickly.
2. Digital sales tools help but only when paired with expertise
Academic research on sales enablement platforms shows performance improves only when tools are combined with skilled human interpretation and cross-functional collaboration.
Dashboards don’t persuade sponsors. People who can interpret data in the context of trial design, site behaviour, and regulatory exposure do.
Automation without insight just accelerates irrelevance.
3. The B2B salesperson’s role has fundamentally changed
Research into the future of B2B sales roles shows a clear shift away from transactional selling toward sense-making, advisory, and consensus-building roles.
In clinical trial technology, the best sellers:
Reframe problems buyers think they’ve already defined
Translate technical capability into operational and regulatory outcomes
Give buyers language they can reuse internally
Make decisions easier after the sales call ends
Buyer intent vs seller behaviour: where deals actually die (and where they live)
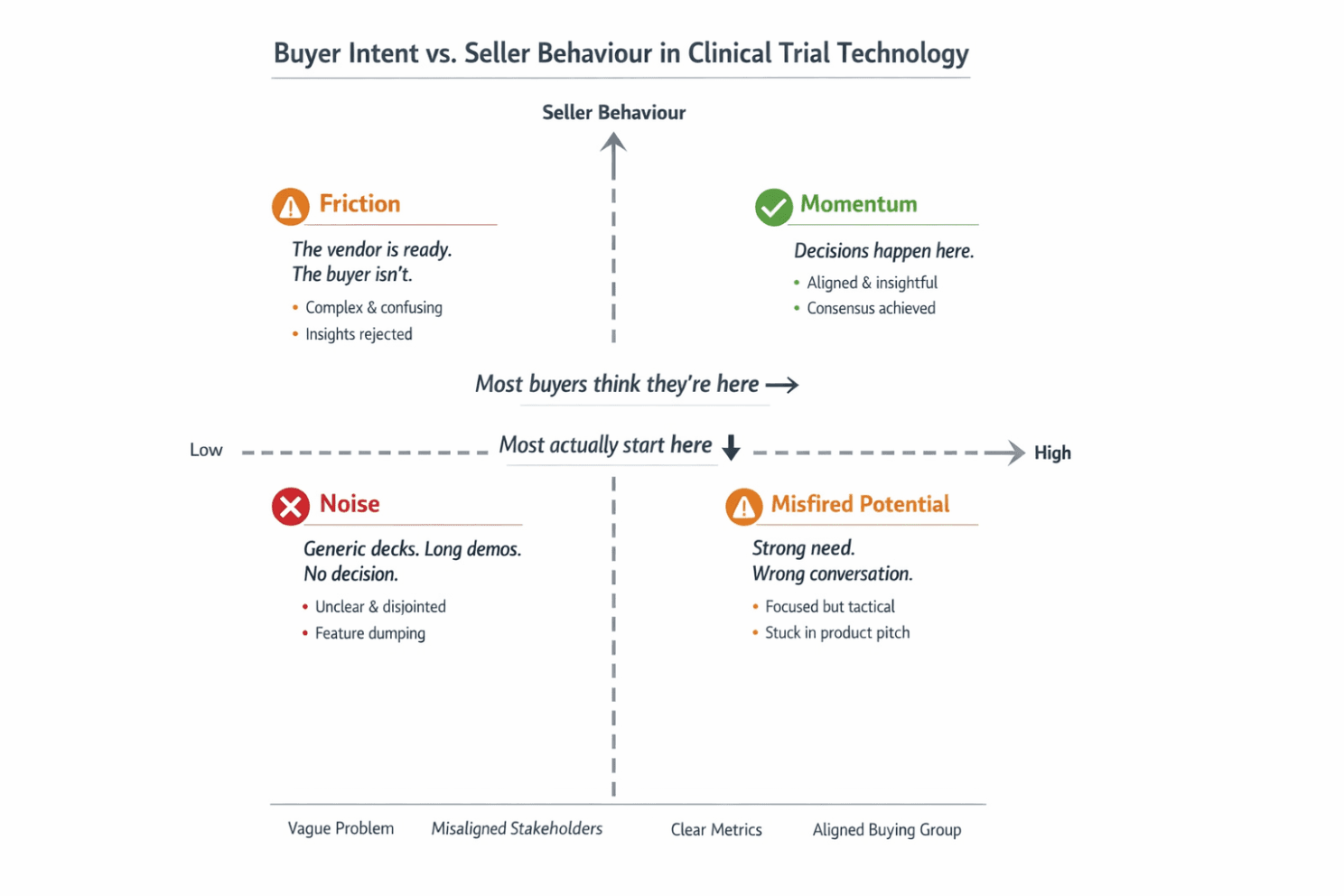
This disconnect between buyer intent and seller behaviour shows up so consistently that it can be visualised.
When buyer clarity is low and sellers default to product-centric messaging, conversations generate noise, not decisions. Momentum only emerges when buyers do the internal work of alignment — and sellers respond with insight that helps organisations reduce risk and reach consensus.
Most stalled clinical trial tech deals don’t fail on capability. They fail on positioning and alignment.
When buyer clarity is low and sellers default to product-centric behaviour, conversations generate noise: polite meetings, long demos and no decisions. When buyers are clear but sellers stay feature-led, differentiation collapses into pricing and procurement.
Momentum only appears when:
Buyers have aligned internally on success criteria
Sellers respond with insight that reframes risk, trade-offs, and outcomes
That is where enterprise clinical trial technology decisions actually happen.
The real divider: successful vs unsuccessful clinical trial tech sellers
Unsuccessful sellers tend to:
Lead with product instead of trial context
Treat “the sponsor” or “the CRO” as a single buyer
Optimise for demos rather than decision pathways
Retreat when QA or Procurement enters
Talk about roadmaps instead of risk reduction
Successful sellers consistently:
Map stakeholder concerns across Clinical, Data, IT, QA, and Procurement
Challenge vague requirements respectfully
Provide narratives buyers reuse in governance forums
Reduce uncertainty more than they create excitement
Design conversations that survive when they’re not in the room
The difference isn’t charm. It’s usefulness under complexity.
A mirror for buyers: how to get better outcomes from vendors
If you’re a sponsor, CRO leader, or clinical operations executive, this should feel uncomfortably familiar.
If buyers want better sales experiences, they also need to buy better.
That means:
Aligning internally before engaging vendors
Defining success beyond “we need a new system”
Rewarding vendors who challenge assumptions
Valuing insight over reassurance
The vendors who push back thoughtfully are often the ones most capable of delivering.
What this really teaches us about B2B sales in 2026
So yes — this is another article about what XYZ has taught me about B2B sales, this time with facts and less fiction.
But the lesson worth keeping isn’t meme-friendly.
In clinical trial technology, the vendors who win aren’t the loudest, fastest, or most automated. They are the ones who help buyers make sense of complexity, reduce risk, and move forward with confidence.
The best clinical trial salespeople don’t close deals. They help buyers close their own internal gaps.
And that’s a lesson worth repeating — even when the memes move on.
References
Singh, J. et al. (2023). The present and future of the B2B sales profession. Journal of Personal Selling & Sales Management. https://www.researchgate.net/publication/369001003_The_present_and_future_of_the_B2B_sales_profession
Rodríguez-Torrico, P. et al. (2025). Sales enablement platforms and digital transformation in B2B sales organisations. Technological Forecasting and Social Change. https://www.sciencedirect.com/science/article/pii/S0040162525002033
Gartner (2022–2024). B2B Buying Behaviour and Buying Group Dynamics. https://www.gartner.com/en/sales/insights/b2b-buying-journey
McKinsey & Company (2023). The New B2B Growth Equation. https://www.mckinsey.com/capabilities/growth-marketing-and-sales/our-insights/the-new-b2b-growth-equation









