>
>
When Your Watch Joins a Clinical Trial: Are Wearables Living Up to the Hype in 2025?
When Your Watch Joins a Clinical Trial: Are Wearables Living Up to the Hype in 2025?
I’m shopping for a new smartwatch. My Garmin Instinct 2 has been my go-to for sport and adventure, but its loud orange casing doesn’t quite pair with a suit. The new Huawei Watch 5, on the other hand, has me hooked: stylish, rugged, and loaded with health features like ECG and the D2 version has blood pressure monitoring.

I’m shopping for a new smartwatch. My Garmin Instinct 2 has been my go-to for sport and adventure, but its loud orange casing doesn’t quite pair with a suit. The new Huawei Watch 5, on the other hand, has me hooked: stylish, rugged, and loaded with health features like ECG and the D2 version has blood pressure monitoring.
And here’s the thought I couldn’t shake: if my watch is already capturing all this data, why isn’t it automatically contributing to clinical research?
That question opened a bigger one: are wearables really being used to their full potential in clinical trials or are we still just scratching the surface?
The Promise of Wearables in Trials
1. Recruitment and Retention Boosts
Wearables reduce patient burden and improve engagement by enabling continuous, real-world monitoring. Instead of multiple site visits, participants can simply wear a device while data flows in passively. This not only lowers dropout rates but also makes recruitment more appealing for underrepresented groups. (npj Digital Medicine)
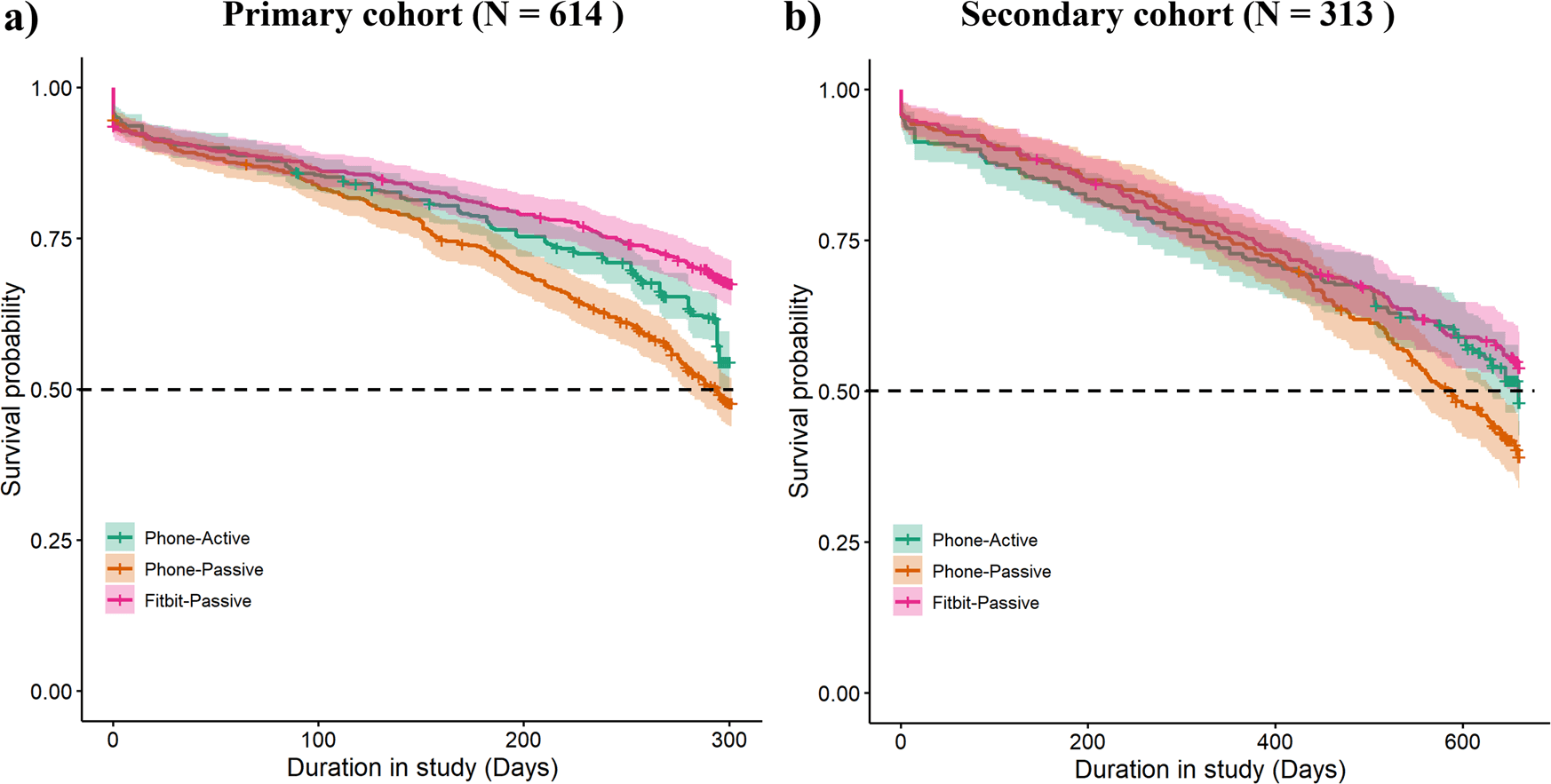
Figure 1: Participant retention curves (Kaplan–Meier) over 43 weeks from the RADAR‑MDD study, showing that passive wearable data (Fitbit‑Passive) achieved ~67.6% retention, compared to ~54.6% (Phone‑Active) and ~47.7% (Phone‑Passive). https://www.nature.com/articles/s41746-023-00890-z
2. Incremental Adoption, Not Full Integration
Despite the promise, most decentralised trials (DCTs) still treat wearables as peripheral add-ons rather than core trial infrastructure. A 2025 review in npj Digital Medicine shows that while adoption is rising, structural integration is still lagging. (npj Digital Medicine)
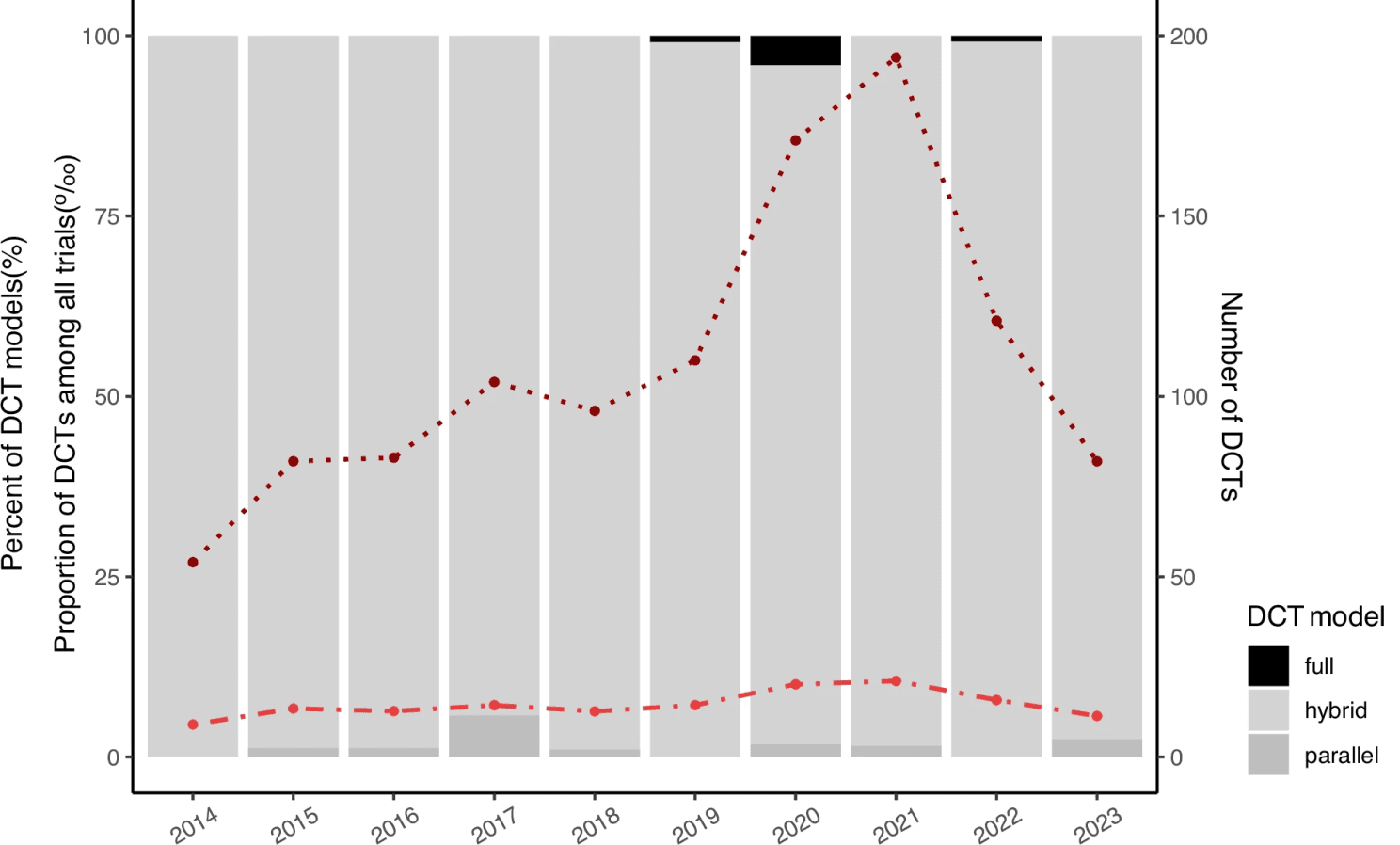
Figure 2: Growth in the use of digital health technologies in clinical trials (2010–2020), broken down by trial design (full, hybrid, parallel), illustrating that true adoption (full DCTs) remains a minority despite overall growth. Reproduced from Jiang et al. (2025), npj Digital Medicine.
3. Remote Data Capture & Digital Biomarkers
A survey in Perspectives in Clinical Research revealed that wearables are one of the most frequently cited tools for remote data capture. Yet, only about half of respondents had ever used them in a trial. Enthusiasm exists, but practical barriers such as infrastructure, validation, and regulatory confidence remain. (Perspectives in Clinical Research)
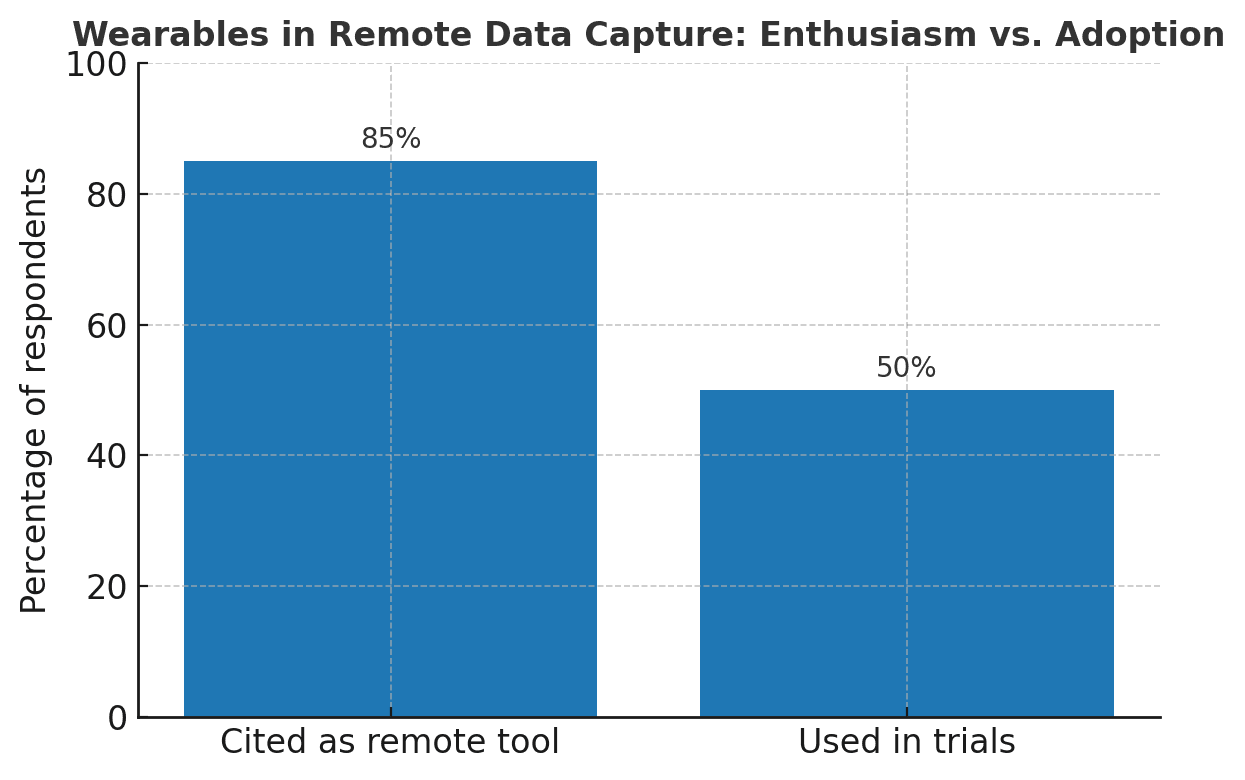
Figure 3: Survey of clinical trial professionals showing widespread recognition of wearables as remote data capture tools (X% citing), yet only ~50% had deployed them in real trials—underscoring the gap between interest and execution (based on Chodankar et al., 2024).
The Validation Challenge
Here’s the elephant in the room: regulators such as the FDA and EMA don’t certify a device just because it looks good on your wrist. They demand fit-for-purpose validation, wearable data must align with gold-standard clinical endpoints.
Challenges include:
Narrow validation cohorts that don’t reflect diverse trial populations.
Device inconsistencies caused by firmware updates, battery issues, and user handling.
Rapid product cycles where new models arrive before the old ones are fully validated.
Until these challenges are systematically addressed, wearables risk staying on the sidelines as “supplementary” tools rather than pivotal sources of trial evidence. (FDA)
A Strategic Solution: Interoperability & Standardisation
The key to unlocking wearable potential isn’t just the device itself, it’s ensuring the data is interoperable, standardised, and regulator-ready.
Adopt open data standards: Frameworks like HL7 FHIR (Fast Healthcare Interoperability Resources) provide a common structure for exchanging wearable data across healthcare systems. (jscdm.org)
Bridge to research models: The FHIR-to-CDISC Mapping Implementation Guide offers a path to convert healthcare data into CDISC formats (CDASH, SDTM), making wearable data usable in regulatory submissions. (jscdm.org)
Leverage industry alliances: Groups like CDISC , TransCelerate BioPharma Inc., and DIA are already building global frameworks to harmonise digital health technologies. Engaging in these collaborations ensures wearable data is not just collected but trusted and reusable. (CDISC overview)
This shift from “novel data streams” to “standardised regulatory endpoints” is the leap needed for wearables to become core trial infrastructure.
Conclusion
My new watch may look sharp in the boardroom, but its greatest value could lie in reshaping the way clinical trials are run. Evidence shows wearables can reduce burden, improve diversity, and create richer datasets.
But in 2025, we’re still scratching the surface. The road to transformation lies in validation, regulatory alignment, and interoperability. When those pieces click, wearables won’t just be good looking gadgets on our wrists, they’ll be trusted engines powering the next era of clinical research.
So next time you glance at your watch, ask yourself: is this just telling the time, or telling the future of trials?
References
Jiang, Y., Leng, Y., Wu, Q., et al. (2025). Understanding the gap between expectations and reality in decentralized clinical trials. npj Digital Medicine, 8:408. https://www.nature.com/articles/s41746-025-01811-y
Chodankar, D., Raval, T. K. (2024). The role of remote data capture, wearables, and digital biomarkers in decentralized clinical trials. Perspectives in Clinical Research. https://pmc.ncbi.nlm.nih.gov/articles/PMC10810055/
Copland, R. R. (2024). The Digital Platform and Its Emerging Role in Supporting Decentralized Trial Activities. Journal of Medical Internet Research, 26:e47882. https://www.jmir.org/2024/1/e47882
U.S. Food & Drug Administration. (2025). Digital Health Technologies (DHTs) for Drug Development. FDA Science & Research. https://www.fda.gov/science-research/science-and-research-special-topics/digital-health-technologies-dhts-drug-development
Ohmann, C., et al. (2024). The FHIR–CDISC Implementation Guide: A Bridge Between Healthcare and Clinical Research. Journal of the Society for Clinical Data Management. https://www.jscdm.org/article/id/162/
Clinical Data Interchange Standards Consortium (CDISC). (2023). Overview of CDISC Standards. https://www.cdisc.org/









